
@ShahidNShah

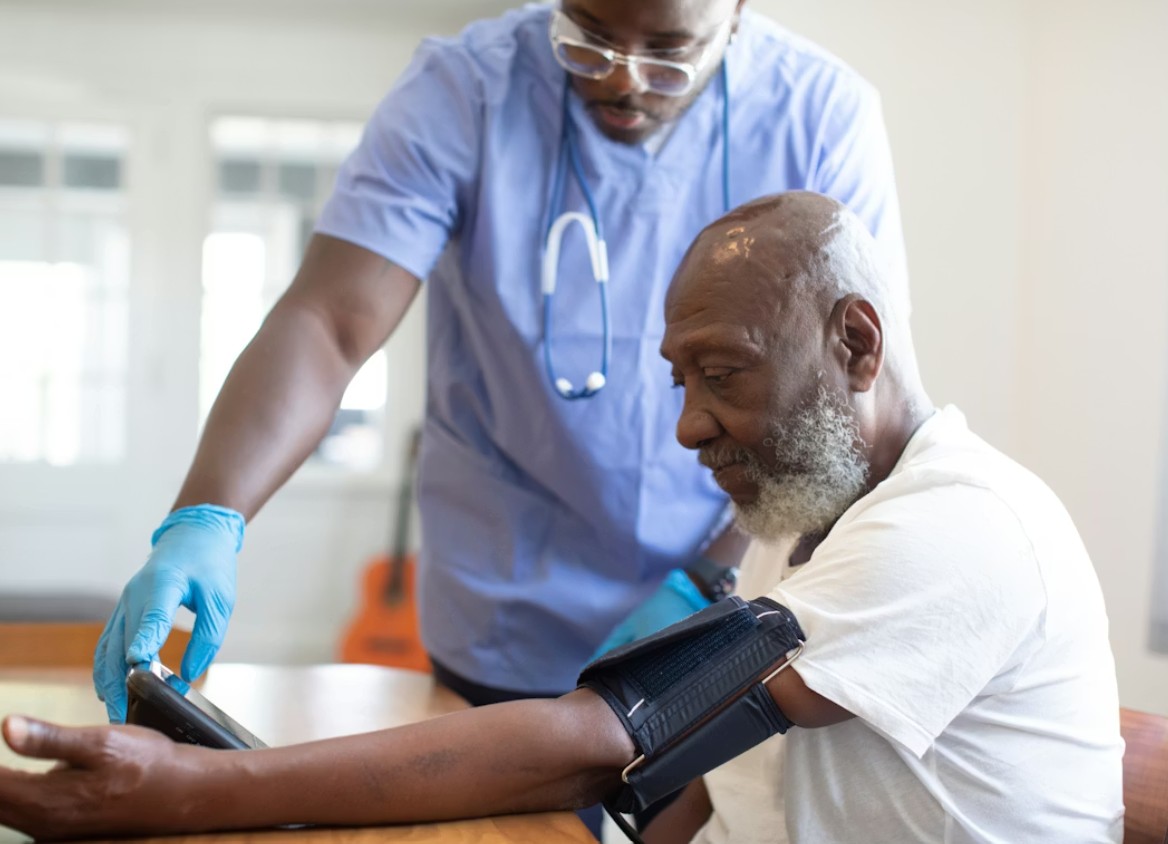
Clinical trials have traditionally been one of the most expensive parts of medical research. Recruiting participants, running physical trial sites, monitoring patients, and managing large amounts of data can make studies slow, complex, and costly.
But this model is starting to change. In recent years, healthcare organizations and research companies have begun using digital tools to simplify different parts of the clinical trial process. Technologies such as remote patient monitoring, AI-assisted recruitment, wearable devices, and paid clinical trials recruiting platforms are helping researchers to access volunteers faster, collect data more efficiently, and reduce the need for in-person visits.
While clinical research is still expensive and highly regulated, many experts believe these changes could gradually lower costs and make trials easier to run in the coming years.
Clinical trials are expensive because they involve far more than testing a treatment. Research teams must recruit participants, run trial sites, monitor patient safety, manage large amounts of data, and meet strict regulatory requirements throughout the study.
Traditional trials also rely heavily on in-person visits. Participants may need to travel regularly to hospitals or research centers for checkups, testing, and follow-up appointments. This increases operational costs and can slow down recruitment, especially for large or long-term studies.
Another major factor is administration. Clinical research generates extensive documentation, reporting, and compliance work, much of which still requires significant human oversight. As trials become more complex and data-heavy, these operational demands continue to grow.
Together, these factors have made clinical trials one of the most time-consuming and costly parts of healthcare innovation.
One of the biggest changes in clinical research is the growth of remote and decentralized trials. Instead of requiring participants to visit research centers for every stage of a study, many newer trials allow at least part of the process to happen from home.
Participants may attend virtual appointments, report symptoms through mobile apps, or use wearable devices that automatically send health data to researchers. In some cases, medications and testing kits can also be delivered directly to patients.
This approach can reduce costs in several ways. Research teams may need fewer physical trial sites, while participants spend less time traveling and are more likely to stay enrolled in the study. Remote models can also help researchers recruit people from wider geographic areas instead of limiting participation to those living near major medical centers.
In the U.S. alone, clinical trials currently recruit participants in over 2,700 cities, highlighting the growing scale and geographic reach of modern clinical research networks.

While decentralized trials are not suitable for every type of study, they are becoming an increasingly important part of modern clinical research.
Artificial intelligence and automation are also starting to change how clinical trials operate. Many research tasks that once required large amounts of manual work can now be completed faster with digital tools.
AI systems can help researchers identify suitable participants by analyzing medical records and matching patients to specific study criteria. Automated platforms can also assist with scheduling, documentation, data processing, and monitoring large datasets during a trial.
These tools may help reduce delays and administrative workload, which are some of the biggest contributors to rising research costs. Faster data analysis and more efficient workflows can also shorten the time needed to complete certain stages of a study.
Many healthcare and biotech companies now outsource parts of clinical research to specialized organizations and global research networks. Instead of managing every stage internally, sponsors often work with external partners that focus on areas such as patient recruitment, data management, regulatory support, or trial monitoring.
This model can help reduce costs by giving companies access to experienced teams and existing research infrastructure without building everything from scratch. Global research networks also allow trials to recruit participants from multiple countries at the same time, which can speed up enrollment and make studies more scalable.
Digital collaboration tools have made this approach even more practical. Research teams in different locations can now share data, monitor progress, and coordinate trial operations remotely.
However, global outsourcing also brings challenges, including differences in regulations, data standards, and communication across regions. As a result, organizations still need strong oversight to maintain consistency and data quality throughout a study.
Several additional changes in healthcare and research may also help lower the cost of clinical trials over time:
Together, these changes may help clinical trials become more efficient and potentially less costly in the coming years.

As the prevalence of osteoarthritis continues to rise globally, clinicians and patients alike are seeking alternatives to traditional treatment pathways that often culminate in joint replacement …
Posted May 13, 2026 Wellness & Prevention
Connecting innovation decision makers to authoritative information, institutions, people and insights.
Medigy accurately delivers healthcare and technology information, news and insight from around the world.
Medigy surfaces the world's best crowdsourced health tech offerings with social interactions and peer reviews.
© 2026 Netspective Foundation, Inc. All Rights Reserved.
Built on Jun 4, 2026 at 3:36pm