
@ShahidNShah

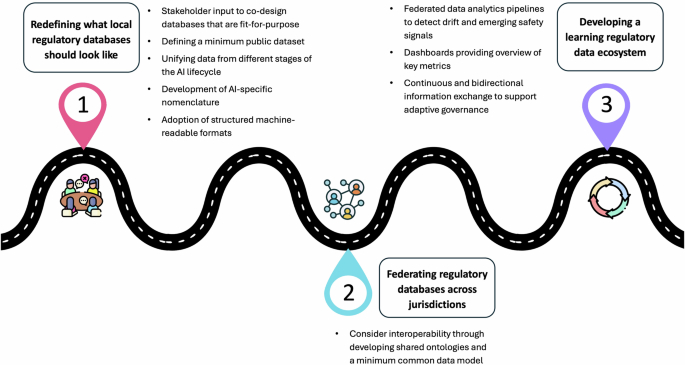
Effective regulatory oversight is a key step in ensuring that artificial intelligence as a medical device (AIaMD) is safe in real-world clinical settings. In this Perspective, we provide insights from our experience working with international regulatory databases, informed by our recent research and the expertise of the multidisciplinary authorship team.
Regulatory databases for AI as a medical device face major challenges in data quality, accessibility, and transparency, limiting effective oversight and safe real-world deployment.
Continue reading at nature.com
AI governance ensures responsible use of AI through rules, processes, and technological tools. Implementing AI governance in healthcare delivery organizations (HDOs) requires understanding necessary …
Connecting innovation decision makers to authoritative information, institutions, people and insights.
Medigy accurately delivers healthcare and technology information, news and insight from around the world.
Medigy surfaces the world's best crowdsourced health tech offerings with social interactions and peer reviews.
© 2026 Netspective Foundation, Inc. All Rights Reserved.
Built on Jun 3, 2026 at 3:35pm