
@ShahidNShah

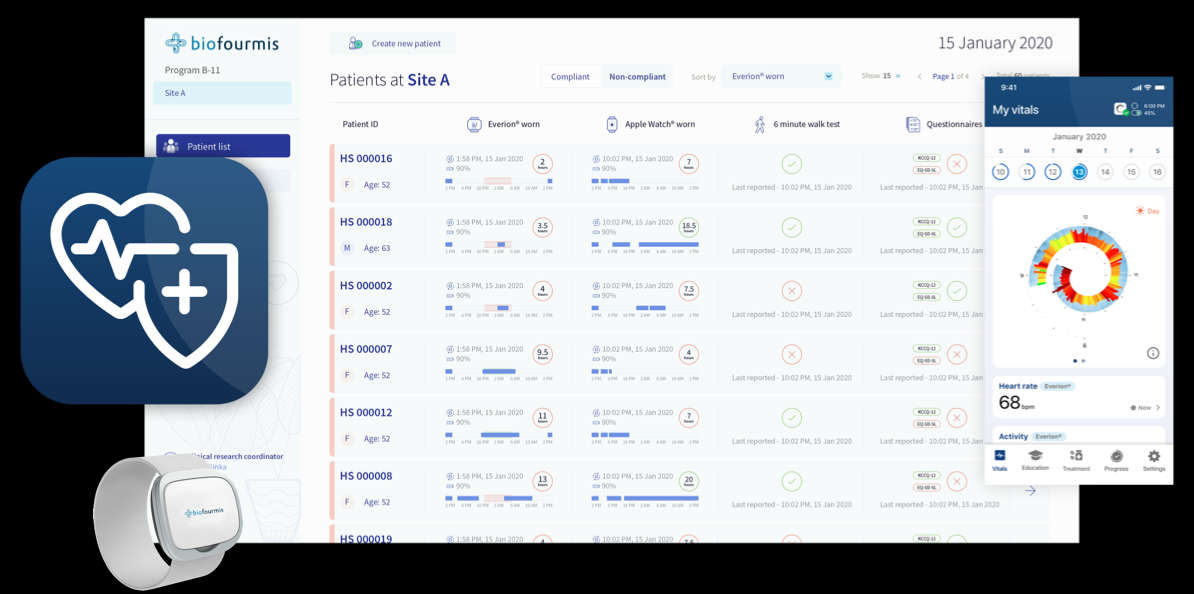
– Biofourmis, a Boston-based global leader in virtual care and digital therapeutics, announced its BiovitalsHF® solution is the first-ever heart failure digital therapeutic to receive a Breakthrough Device designation from the U.S. Food and Drug Administration (FDA).
– The Breakthrough Devices Program was created to provide patients and providers with timely access to medical devices (in this case, software as a medical device, or SaMD) by speeding up their development, assessment, and review while preserving the statutory standards for premarket approval, 510(k) clearance, and De Novo marketing authorization, according to the FDA.
Continue reading at hitconsultant.net
Despite investor fears of a telehealth slowdown, the market banked a record-setting $5 billion in 163 funding deals in the second quarter of 2021. Global telehealth investment rose for the fourth …
Posted Aug 2, 2021 Fundraisers Telehealth
Connecting innovation decision makers to authoritative information, institutions, people and insights.
Medigy accurately delivers healthcare and technology information, news and insight from around the world.
Medigy surfaces the world's best crowdsourced health tech offerings with social interactions and peer reviews.
© 2024 Netspective Media LLC. All Rights Reserved.
Built on Apr 19, 2024 at 8:35am